Roquette Pyrogen-Free Range of Products for Parenteral Preparations (Injection, Infusion and Dialysis Solutions)
Published June 08, 2026
Authors
Blouet Elham
Pharm.D. - Head of Marketing|Biopharma - Injectables, Dialysis and Specialty APIs, Roquette
Introduction
Parenteral preparations (injection, infusion, dialysis…) must be, among other requirements, sterile and “pyrogen-free.” Ingredients used (active pharmaceutical ingredients, excipients) must be of high-quality standards in terms of physicochemical and microbial purity.
Pyrogenic substances that cause fever can be either an endotoxin or an exotoxin, although most pyrogens are endogenous. Endotoxins are lipopolysaccharide (LPS) molecules in part of the cell wall of gram-bacteria and released primarily upon cell lysis. Endotoxins are tested with the in-vitro LAL test (Limulus Amoebocyte Lysate).
Materials and Key Technical Points
LYCADEX® PF DEXTROSE MONOHYDRATE PYROGEN-FREE: LIFESAVING MOLECULE
Dextrose monohydrate pyrogen-free for injectable solutions (injection and infusion solutions), dialysis solutions and parenteral nutrition.
LYCADEX® PF is manufactured in two Roquette sites: Lestrem, France, and Keokuk, Iowa, USA.
Quality Assurance
• Dedicated manufacturing line with controlled environment (no cross-contamination)
• Purification steps (high quality)
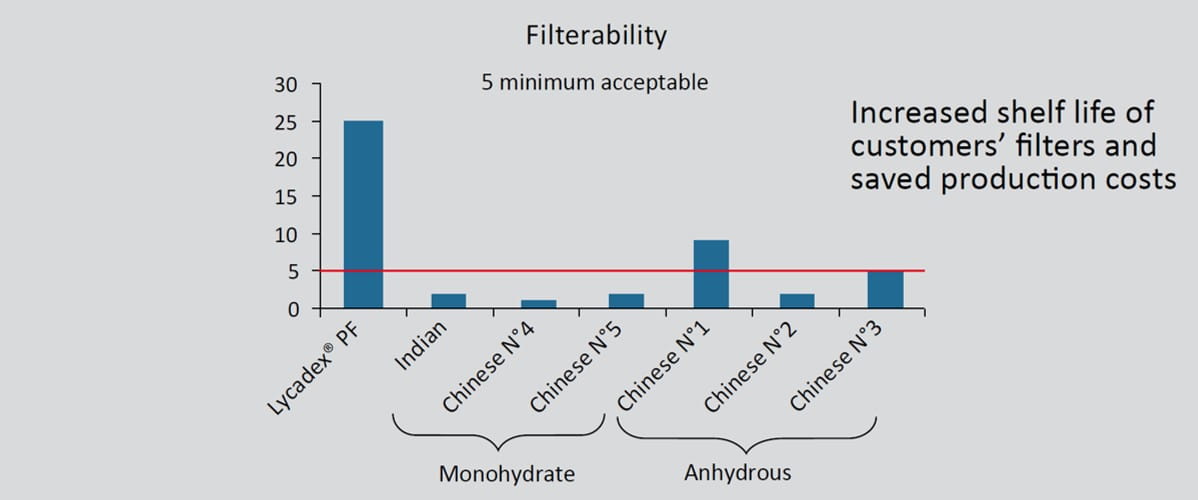
• Improved filterability; customer’s benefit
• In-process controls (for example, LAL) and end-product control for endotoxins: LAL test
• Compliance: US cGMP + EU GMP part II, (ICH Q7)
Regulatory Aspects
• Compliance to EP, USP, JP, IP and ChP
- CEP (Keokuk and Lestrem)
- US-DMF type II (Keokuk and Lestrem)
- Chinese DMF: Y20190000012
• Approved by Competent Authorities such French Health Authorities (ANSM) and U.S. FDA.
• GMP (EU GMP part II, ICHQ7) Certificate
Key technical points: filterability
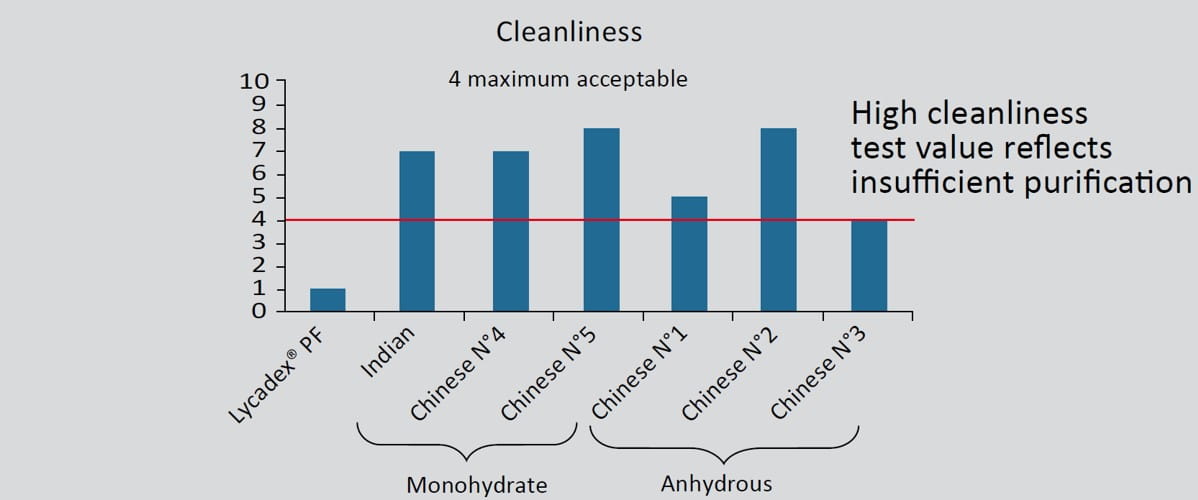
Key technical points: cleanliness
OTHER PYROGEN-FREE SOLUTIONS
Quality Assurance Key Points
| Quality Assurance PEARLITOL® PF Mannitol NEOSORB® PF Sorbitol KLEPTOSE® HPB, HP parenteral grade Sodium Gluconate Pharma KLEPTOSE® HPB-LB parenteral grade | • Campaign basis production; controlled environment (no cross-contamination) • Purification steps (high quality) • In-process controls and end-product testing • LAL test (endotoxins testing) • Compliance to GMP (ICH Q7) |
Regulatory Aspects
| REGULATORY ASPECTS LYCADEX® PF |
|
| REGULATORY ASPECTS PEARLITOL® PF Mannitol NEOSORB® PF Sorbitol |
|
| REGULATORY ASPECTS KLEPTOSE® HPB, HP parenteral grade |
|
| REGULATORY ASPECTS KLEPTOSE® HPB-LB parenteral grade |
|
| REGULATORY ASPECTS Sodium Gluconate Pharma |
|
Conclusion
Our pyrogen-free/endotoxins controlled range of products and our vertically integrated portfolio support our customers’ needs for supply chain transparency and traceability. We understand our customers’ concerns and need for compliance to ensure patient safety. Additionally, we offer expertise in developing a variety of custom solutions, partnering with you from the earliest development stages of a project.
Disclaimer
®Registered trademark(s) of Roquette Frères.
The information contained in this document is to the best of our knowledge true and accurate, but all instructions, recommendations or suggestions are made without any guarantee. Since the conditions of use are beyond our control, we disclaim any liability for loss and/or damage suffered from use of these data or suggestions. Furthermore, no liability is accepted if use of any product in accordance with these data or suggestions infringes any patent. No part of this document may be reproduced by any process without our prior written permission. For questions about a product’s compliance with additional countries’ standards not listed above, please contact your local Roquette representative.