Microcrystalline Cellulose (MCC) 102 and 200 Screening as Filler in Direct Compression for Acetaminophen and Griseofulvin as Model Drugs
Published April 02, 2026
Presented at the 13th World Meeting on Pharmaceutics, Biopharmaceutics and Pharmaceutical Technology, March 28-31, 2022, Rotterdam, Netherlands
Authors
Dongyue Yu
University of Maryland Baltimore
Stephen W. HOAG
University of Maryland Baltimore
Dr Carmen Popescu
Global Technical Applications Specialist, Roquette
Introduction
The oral route of administration is the most common and preferred method of delivery. Tablets have many advantages as a dosage form, like high patient compliance, portability, relatively low cost, and convenient scaling-up. Microcrystalline cellulose (MCC) is one of the most commonly used fillers. In this study, four commercially available MCC 102 and 200 were used. Acetaminophen (APAP), a BCS Class III compound, and griseofulvin (GSF), a BCS Class II drug, were used as model drugs. The goal of this study is to evaluate each component to obtain a suitable formulation.
Materials and Methods
Materials :
Acetaminophen (APAP) (PH EUR/USP 90% DC/C; batch no: D2803164) was manufactured by ATABAY (İstanbul, Turkey). Griseofulvin (GSF) USP was obtained from Letco (Letco Medical, L.L.C.; Wayne, PA). MCCs 102 and 200 were as follows: MCC1 (AVICEL® PH), MCC2 (MICROCEL® SD), MCC3 (MICROCEL® MC) and MCC4 (VIVAPUR®)
Methods :
Tablets were made using 11.28 mm diameter (1cm2) round, flat-faced TSM-D tooling with a target weight of 250 mg for all samples using a Styl’One Evolution simulating Korsch XL-400 rotary compressor.
Dissolution:
The paddle method (apparatus 2) was used at 50 rpm. Tablets were made under 15 kN compression force at medium compression speed (35 rpm = 1015 tab/min, simulating Korsch XL-400 rotary compressor). Drug release concentration was evaluated by HPLC based on the standard calibration API curve.
UPLC methods:
For APAP, the mobile phase is a mixture of phosphate buffer (pH = 4.88) and methanol (80:20, v/v). The flow rate was set at 0.8 mL/min, and the injection volume was 10 μL, the wavelength was 242 nm. For GSF, the mobile phase is a mixture of 20 mM aqueous sodium dihydrogen phosphate solution and acetonitrile (55:45, v/v). The final pH of the mobile phase is adjusted to 3.5 with phosphoric acid. The flow rate was set at 1.0 mL/min and the injection volume was 10 μL. GSF fluorescence measurements are run at excitation wavelengths of 300 nm. The mobile phase is filtered through a 0.45 μm GHP membrane filter and degassed before use.
API content uniformity study was performed as per UPS <905> requirements.
RESULTS
1. Lubricant concentration screening:
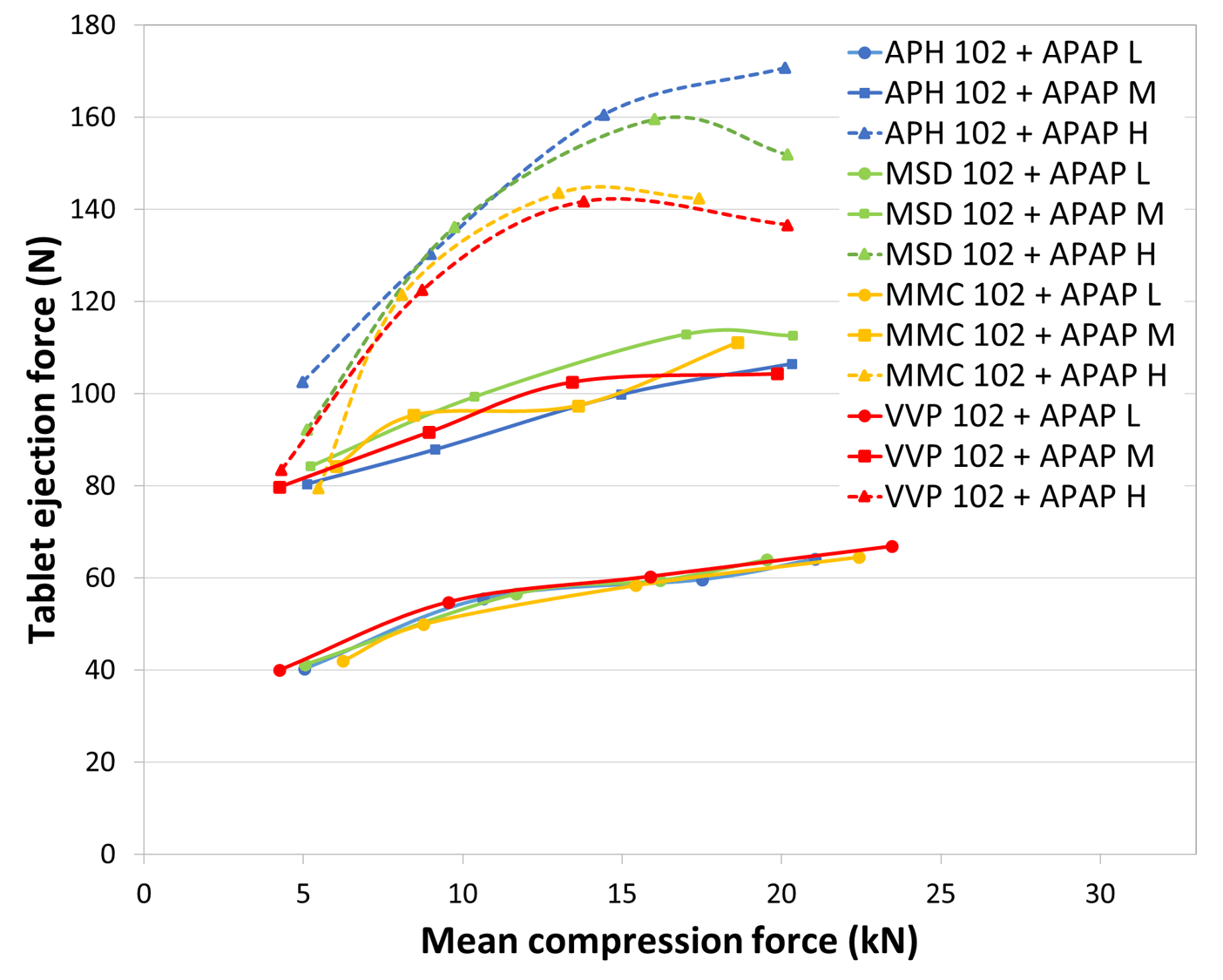
Optimal lubricant concentration, 0.5% of Mg Stearate, was selected based on preliminary screening and lead to ejection force under 200 N for all MCC brands and grades, indicating a good lubrication performance (figure 1) during tablet compaction.
Figure 1. Ejection force of APAP-MCC 102 formulations (40% APAP + 59.5% MCC + 0.5% Mg Stearate) simulating Korsch XL-400 rotary compressor (L = 1 rpm = 2100 tab/h, M = 35 rpm = 73500 tab/h, H = 70 rpm = 147000 tab/h)
2. Dilution potential of MCCs as a filler/binder in direct compression (DC):
2.1 MCC dilution potential of acetaminophen (APAP)
Preliminary tests were performed with 30%, 40%, and 50% of APAP loading in the presence of 0.5% of Mg Stearate under compression forces between 5 kN to 30 kN. However, lamination of the tablet with 50% APAP load was observed after compression. Therefore, we decided to use 40% of APAP load as a formulation model for the compaction study.
2.2 MCC dilution potential of griseofulvin (GSF) in DC
Preliminary screening of 20%, 30%, and 40% of GSF load in the presence of 0.5% of Mg Stearate under compression forces between 5 kN to 30 kN. Capping was observed for tablets at 40% GSF load in the presence of MCC 200 at high compression pressures (20 kN and 30 kN). Based on this preliminary result, we decided to run the formulation at 35% GSF load, and consequently, we did not detect any defects in the resulting compacts at high pressure. Both APAP and GSF showed poor flowability. However, all brands and grades of MCC were able to compensate this flow issue and generate good tablets.
3. Disintegrant concentration screening:
Tablets with and without two disintegrants GLYCOLYS® sodium starch glycolate (Roquette), SOLUTAB® A croscarmellose sodium (Roquette) at 5% were compressed at 15 kN and 20 kN. We understood that these formulations do not require any disintegrant as long as the disintegration time is less than 60 seconds without disintegrant and less than 40 seconds in the presence of disintegrant.
4. Drug content and content uniformity
According to USP APAP monograph, the APAP unit (formula as per table 1) should contain not less than 90% and not more than 110% of the labeled amount of APAP. In our case, the measured amount of APAP should be within the range of 36 mg – 44 mg per 100 mg. Meanwhile, the GSF tablet should contain not less than 90% and not more than 115% of the labeled amount of GSF.
Table 1. Formulation components
| Component | Percentage | Component | Percentage | |
| API | APAP | 40 % | GSF | 35 % |
| Filler/binder | MCC | 59.50 % | MCC | 64.50 % |
| Lubricant | Mg Stearate | 0.50 % | Mg Stearate | 0.50 % |
In our case, the measured amount of GSF should be within the range of 31.5 mg – 40.25 mg per 100 mg. All the formulations attain the USP requirement (table 2).
Table 2. Drug content study of APAP and GSF tablets with MCCs
| APAP Amount/100 mg | GSF Amount/100 mg | |||
| Mean | SD | Mean | SD | |
| MCC1 102 | 40.4 | 0.4 | 34.2 | 0.6 |
| MCC2 102 | 41.6 | 1.0 | 34.4 | 0.8 |
| MCC3 102 | 41.0 | 0.0 | 35.1 | 0.6 |
| MCC4 102 | 41.1 | 1.2 | 35.4 | 0.1 |
| MCC1 200 | 39.9 | 0.1 | 34.6 | 0.5 |
| MCC2 200 | 40.8 | 1.0 | 34.5 | 0.9 |
| MCC3 200 | 41.7 | 0.2 | 34.4 | 0.4 |
| MCC4 200 | 40.8 | 0.6 | 34.8 | 0.1 |
Drug release percentage is calculated using the measured concentration API value in the dissolution medium divided by the amount of API formulated in the tablet. Dissolution evaluation was performed using two different runs to prove data consistency (table 3 shows an example of the APAP % released at different time points). All the tablets with MCC 102 were able to release more than 80% of the tablet amount of APAP in 30 min.
Table 3. Drug dissolution of APAP and GSF tablets with MCCs
| APAP Tablets | GSF Tablets | |||||
| 30 min | 60 min | 90 min | 30 min | 60 min | 90 min | |
| MCC1 102 | 80.7 % | 91.7 % | 95.1 % | 89.3 % | 97.4 % | 98.0 % |
| MCC2 102 | 83.1 % | 98. % | 99.7 % | 82.6 % | 91.2 % | 93.3 % |
| MCC3 102 | 94.2 % | 97.4 % | 100.0 % | 79.0 % | 94.0 % | 95.8 % |
| MCC4 102 | 84.8 % | 94.5 % | 98.4 % | 46.4 % | 67.8 % | 75.1 % |
| MCC1 200 | 81.1 % | 91.0 % | 97.6 % | 64.1 % | 77.0 % | 79.8 % |
| MCC2 200 | 92.8 % | 98.9 % | 99.2 % | 87.7 % | 94.3 % | 95.5 % |
| MCC3 200 | 85.8 % | 95.8 % | 99.7 % | 70.6 % | 82.0 % | 84.4 % |
| MCC4 200 | 81.5 % | 93.1 % | 97.8 % | 77.5 % | 84.0 % | 84.7 % |
Content uniformity is an important quality parameter of final solid dosage compact. As per table 4, all the acceptance values are less than the maximum allowed acceptance value of 15.0. GSF formulations have relatively higher acceptance values (AV) compared with APAP formulations due to significant difference between the particle size of GSF and MCCs.
Table 4. Values of weight variation of APAP and GSF formulations
| AV of APAP tablets | AV of GSF tablets | |
| MCC1 102 | 3.47 | 5.62 |
| MCC2 102 | 3.04 | 5.31 |
| MCC3 102 | 2.63 | 3.6 |
| MCC4 102 | 4.3 | 3.24 |
| MCC1 200 | 4.89 | 8.17 |
| MCC2 200 | 4.96 | 8.98 |
| MCC3 200 | 5.75 | 8.61 |
| MCC4 200 | 5.97 | 6.71 |
Conclusion
In support of the formulator flexibility using MCC (GRADE 102 and 200) as a filler in DC, we can conclude that despite individual powder properties differences the final criteria that qualify them as equivalent is dissolution profile and content uniformity performance. Both 102 and 200 MCC grades of all tested manufacturers lead to disintegration time less than 1 minute, 80% of drug dissolution within 30 min and USP content uniformity conformity.
References
Reier, G. E. and Shangraw, R. F.. “Microcrystalline cellulose in tableting.” Journal of Pharmaceutical Sciences 55, 5 (1966): 510-514.
https://doi.org/10.1002/jps.2600550513
AVICEL® is a registered trademark of FMC, Germany.
VIVAPUR® is a registered trademark of JRS Pharma, Germany
MICROCEL® is a registered trademark of Roquette Frères in Benelux, Brazil, Canada, Chile, France, Germany, Italy, Mexico, the United Kingdom, and the United States of America and is pending in other countries or regions.
® Registered trademark(s) of Roquette Frères.
The information contained in this document is to the best of our knowledge true and accurate, but all instructions, recommendations or suggestions are made without any guarantee. Since the conditions of use are beyond our control, we disclaim any liability for loss and/or damage suffered from use of these data or suggestions. Furthermore, no liability is accepted if use of any product in accordance with these data or suggestions infringes any patent. No part of this document may be reproduced by any process without our prior written permission. For questions about a product’s compliance with additional countries’ standards not listed above, please contact your local Roquette representative.