SWEETPEARL® P 300 DC - MALTITOL
- Directly compressible
Mean particle diameter : 300 μm. Can be supplied with food grade documentation for nutraceuticals.
Applications
- 1. Solid Forms - Tablets
- Chewable Tablets
- Swallowable Tablets
- Effervescent Tablets
Functional properties
- Formulations Aids
- Fillers and Binders for Direct Compression
- Fillers and Binders for Roller Compaction
- Sensory Enhancers
- Sweetening agent
Physical and chemical properties
- General Properties
- Multicompendial
Documents
Product Specification Sheet
Name
Region
Size
Download
SWEETPEARL® P300 DC
478,05 Ko
Region :
478,05 Ko
Safety Data Sheet
Name
Region
Language
Size
Download
SWEETPEARL® P300 DC
Europe, BE
EN
521,35 Ko
Region : Europe, BE
EN
521,35 Ko
SWEETPEARL® P300 DC
Europe, CH
EN
521,00 Ko
Region : Europe, CH
EN
521,00 Ko
SWEETPEARL® P300 DC
Europe, DE
EN
523,72 Ko
Region : Europe, DE
EN
523,72 Ko
SWEETPEARL® P300 DC
Europe, FR
EN
521,04 Ko
Region : Europe, FR
EN
521,04 Ko
SWEETPEARL® P300 DC
Europe, GB
EN
384,12 Ko
Region : Europe, GB
EN
384,12 Ko
SWEETPEARL® P300 DC
Oceania, AU
EN
270,42 Ko
Region : Oceania, AU
EN
270,42 Ko
SWEETPEARL® P300 DC
Asia, IN
EN
268,57 Ko
Region : Asia, IN
EN
268,57 Ko
SWEETPEARL® P300 DC
Europe, IT
EN
523,55 Ko
Region : Europe, IT
EN
523,55 Ko
SWEETPEARL® P300 DC
Asia, MY
EN
271,24 Ko
Region : Asia, MY
EN
271,24 Ko
SWEETPEARL® P300 DC
Europe, BE
DE
533,68 Ko
Region : Europe, BE
DE
533,68 Ko
SWEETPEARL® P300 DC
Europe, BE
FR
634,61 Ko
Region : Europe, BE
FR
634,61 Ko
SWEETPEARL® P300 DC
Europe, BE
NL
529,12 Ko
Region : Europe, BE
NL
529,12 Ko
SWEETPEARL® P300 DC
Europe, CH
DE
534,06 Ko
Region : Europe, CH
DE
534,06 Ko
SWEETPEARL® P300 DC
Europe, CH
FR
635,03 Ko
Region : Europe, CH
FR
635,03 Ko
SWEETPEARL® P300 DC
Europe, CH
IT
622,53 Ko
Region : Europe, CH
IT
622,53 Ko
SWEETPEARL® P300 DC
Europe, CZ
CS
653,79 Ko
Region : Europe, CZ
CS
653,79 Ko
SWEETPEARL® P300 DC
Europe, DE
DE
538,18 Ko
Region : Europe, DE
DE
538,18 Ko
SWEETPEARL® P300 DC
Europe, DK
DA
530,61 Ko
Region : Europe, DK
DA
530,61 Ko
SWEETPEARL® P300 DC
Europe, ES
ES
535,29 Ko
Region : Europe, ES
ES
535,29 Ko
SWEETPEARL® P300 DC
Europe, FR
FR
634,79 Ko
Region : Europe, FR
FR
634,79 Ko
SWEETPEARL® P300 DC
Americas, Asia, Oceania, Africa
RU
416,53 Ko
Region : Americas, Asia, Oceania, Africa
RU
416,53 Ko
SWEETPEARL® P300 DC
Americas, Asia, Oceania, Africa
PT
285,62 Ko
Region : Americas, Asia, Oceania, Africa
PT
285,62 Ko
SWEETPEARL® P300 DC
Europe, IT
IT
624,38 Ko
Region : Europe, IT
IT
624,38 Ko
SWEETPEARL® P300 DC
Asia, JP
JA
399,41 Ko
Region : Asia, JP
JA
399,41 Ko
SWEETPEARL® P300 DC
Asia, KR
KO
323,62 Ko
Region : Asia, KR
KO
323,62 Ko
Get in touch with our Technical Experts
Please feel free to contact our technical experts for support during the development process.
Technical data
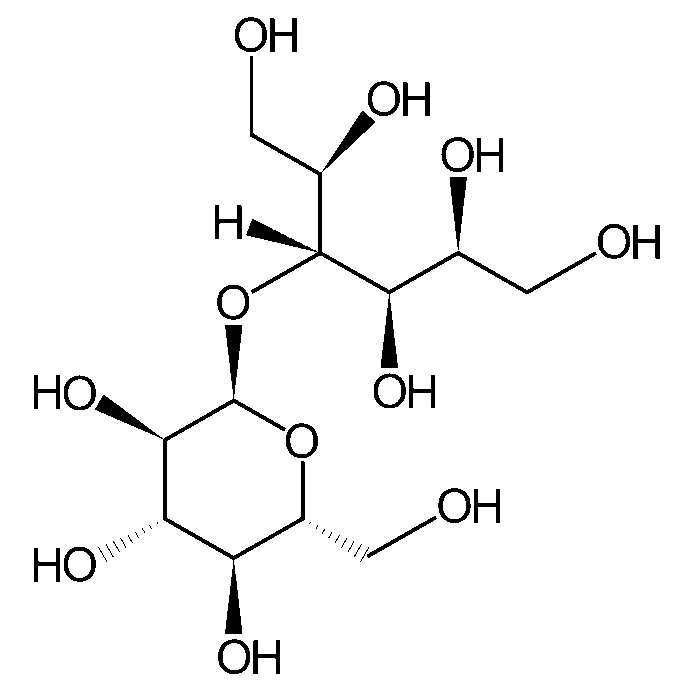
| Synonyms | D-Maltitol |
|---|---|
| CAS number | 585-88-6 |
| Physical form or apperance | White or almost white crystalline powder |
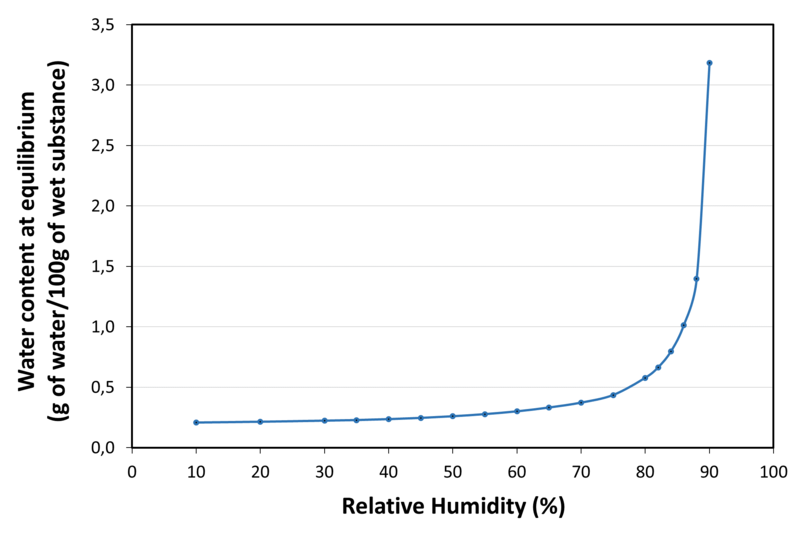
| Application | SweetPearl® P300 DC maltitol is a direct compression excipient with high chemical stability and low hygroscopicity. It is suitable for chewable, swallowable or effervescent tablets. It is a non-cariogenic and non-acidogenic sugar-free sweetener. |
| Teste/Odor | Sweet (sweetening power about 90% that of sucrose) |
| Morphology |
|
| Water sorption isotherm at 20°C |
|
| Chemical Structure |
|
| Average molecular weight | 344.3 g/mol |
| Nominal Water content (LOD) | 0.30 |
| Maximal Water content (LOD) | 1.00 |
| Solubility | Freely soluble in water (153 g/100 ml at 20°C), practically insoluble in anhydrous ethanol |
| Minimum melting temperature | 148 °C |
| Maximum melting temperature | 151 °C |
| Average mean particle diameter | 300 |
| dv10 Particle size distribution | 130 |
| dv50 Particle size distribution | 300 |
| dv90 Particle size distribution | 410 |
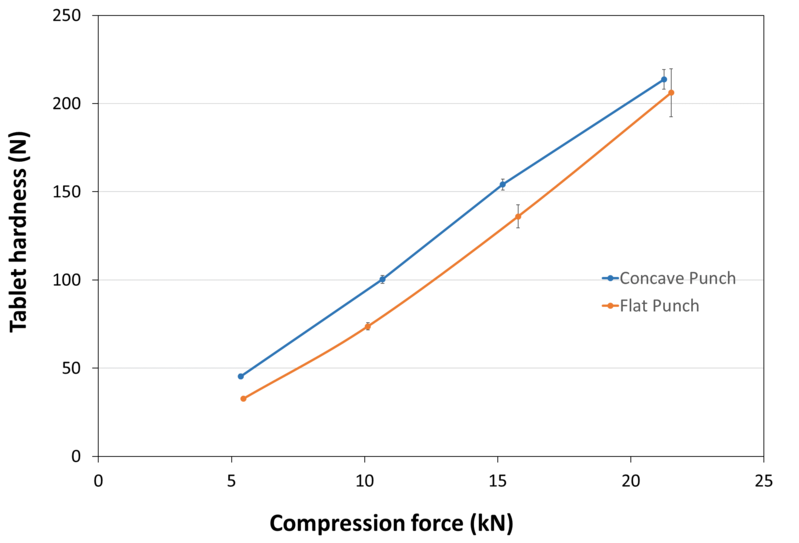
| Tablet Hardness |
|
| Experimental Conditions for Compression Behavior_Tablet Press | STYLCAM 200R |
| Experimental Conditions for Compression Behavior_Production Speed | 10 tabs/min (linear punch velocity: 38 mm/s ; simulated rotary press speed: 60000 tablets/hour) |
| Experimental Conditions for Compression Behavior_Tooling | Diameter 10 mm R9 concave and diameter 10 mm flat |
| Experimental Conditions for Compression Behavior_Formuls | 98.8% SweetPearl® P300 DC / 1.2% magnesium stearate (Concave punch)99.4% SweetPearl P300 DC / 0.6% magnesium stearate (Flat punch) |
| Experimental Conditions for Compression Behavior_Tablet Mass | 455 mg (Concave punch)400 mg (Flat punch) |
| Powder Flowability (according to Ph.Eur. 2.9.16, 10mm outflow opening) | 7 |
| Bulk Density (g/ml) | 0.59 |
| Tapped Density (g/ml) | 0.71 |
| True Density (g/ml) | 1.62 |
| Specific Surface Area (m²/g) | 0.19 |
| Angle of Repose (°) | 31 |