Starch-based excipients in continuous wet granulation applications
Published June 04, 2026
Author(s)
Olaf Haeusler
Pharm.D. - Global Technical Application Specialist, Roquette
Introduction
The future of pharmaceutical manufacturing industry is certainly linked with continuous manufacturing processes. Replacing the common batch procedures permits a much higher control of the product quality, combined with economic advantages such as reduced production costs and shorter production times. However, moving from traditional processes toward continuous ones, requests new technical solutions and selected excipients with adapted properties, potentially providing new functionalities.
The twin-screw granulation (TSG) is the continuous granulation technology providing a flexible approach to design granules. It requires a minimum effort for scale up and can easily be adjusted to the needs of the continuous line. A twin-screw granulator is a regime-separated granulator. This means that the granulation process steps (i.e., nucleation, coalescence, consolidation and breakage) occur physically separated from each other along the length of the screw. There are many direct and indirect process variables to modulate the granulation step and to modify the properties of the obtained granules. These process parameters are for example the powder throughput, the barrel fill level but also the screw configurations and the used Liquid/Solid ratio (L/S ratio). Generally, the particle size of the obtained granules increases with the used L/S ratio, using either high shear batch granulation or continuous TSG. However, the continuous twin-screw granulation is less sensitive to liquid amount than high-shear granulation when comparing the granule size increase. Granules obtained with the TSG “grow” in a much more defined environment, offering a better handling of the whole granulation process. In general, the tablets made of TSG granules have a higher tensile strength than tablets made of high shear granules. Continuous twin-screw granulation is therefore a good technical alternative for high-shear granulation (batch process).
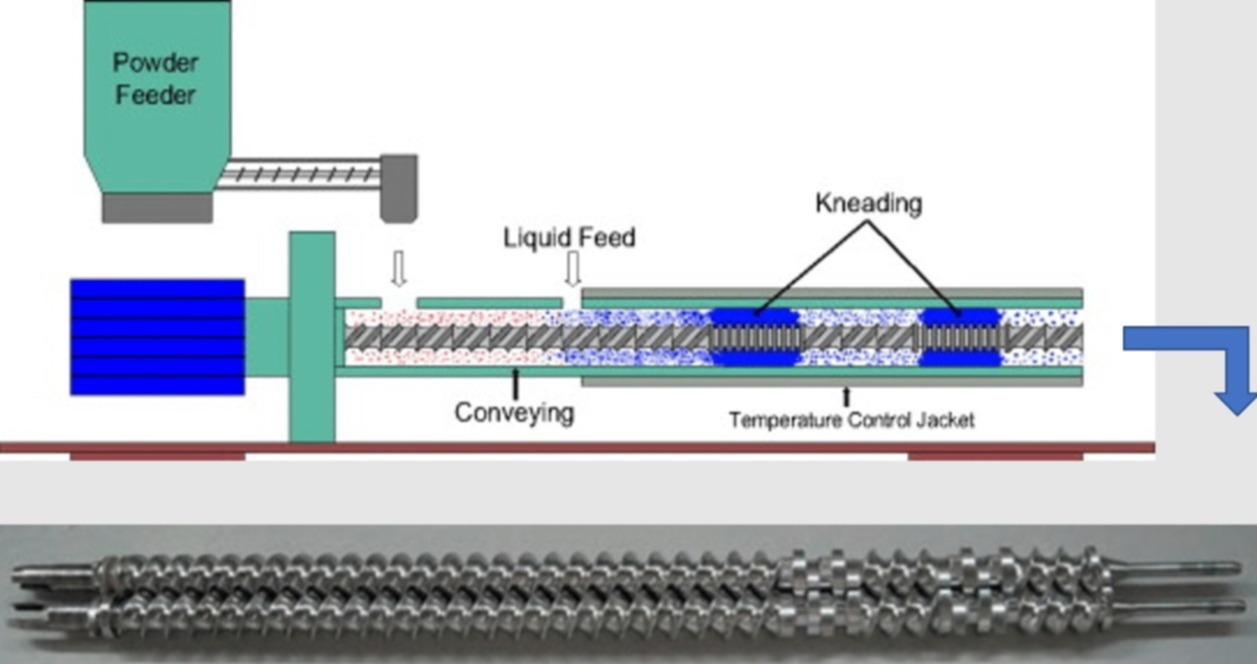
Figure 1: schema of continuous wet granulation and picture of a typical screw configuration (co-rotating screws) with conveying elements and kneading elements (right).
Comparing the batch and the continuous granulation shows major process differences. The very short residence time in the granulating tool (5 – 20 seconds) in continuous wet granulation (CWG) is important, combined with the use of rather low amounts of wetting fluids. Binder, usually added in powder form, needs specific attributes to become very rapidly wetted and hydrated (to activate its binding properties). Therefore, not all traditional wet granulation (WG) binders perfectly fit with these new process needs. Other excipients and drug substances influence the granulation process as well, e.g., due to partial dissolution.
Role of bulk excipients
Common filler-binders (such as dicalcium phosphate, lactose, mannitol and microcrystalline cellulose [MCC]) play a major role in the granulation process by modulating the quality of the obtained granules (and tablets made thereof). This is linked with differences in their physical and chemical properties such as their wettability, their solubility in the granulation liquid or the possible formation of amorphous phases.
The partial excipient dissolution could enhance the granulation result. Its recrystallization during drying is useful to optimize the granules’ texture for tableting. For example, mannitol can crystallize again when drying by forming smaller crystals and crystallites, giving the granules a higher surface area than the primary powder leading to better compaction properties. Literature reports a decrease of the tableting of MCC, when wetted with water during granulation. This undesirable effect could be limited by adding mannitol, helping to recover an acceptable porosity.
Wet granulation binder characterisation and criteria of its choice
The physical and chemical properties of the binder polymer determine the granulation process and the quality of the obtained granules and tablets. A prior binder characterization facilitates the binder selection to design the best possible formulation.
The first key factor for successful granulation process is the binder’s wetting capacity of hydrophobic surfaces (example ibuprofen), mandatory for an effective bond formation between all particles. In addition, the fast binder activation within a very short time depends on effective interactions between the binder and water. A less wettable binder requests typically higher L/S ratio for comparable performance. Slower binder activation correlates with highly viscous binders having slow dissolution kinetics.
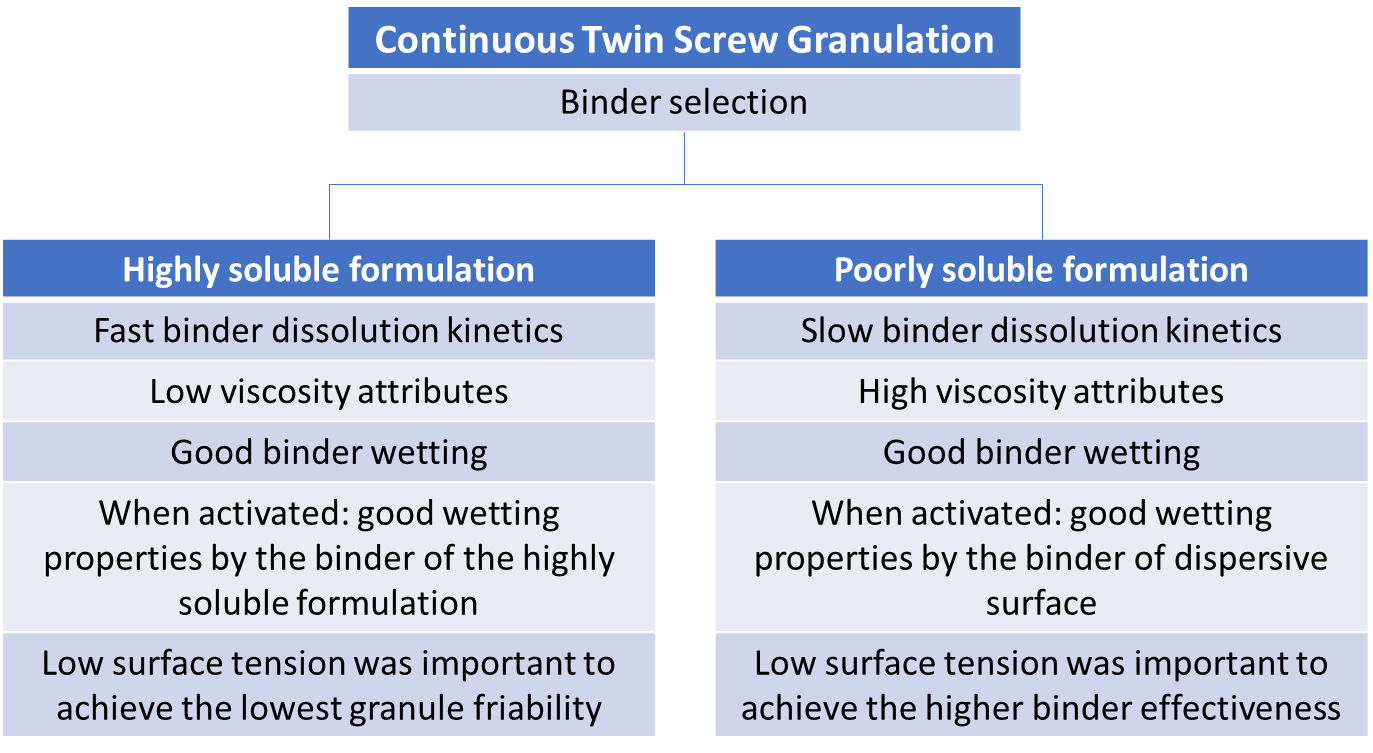
Furthermore, because of their solubility, powder blends request different binder properties. On the one hand, highly soluble formulations demand for a fast binder activation (correlated with its fast dissolution kinetics, low viscosity and good wetting properties). On the other hand, in poorly soluble formulations, the use of binder developing higher viscosity and having slower dissolution kinetics results in better granules and tablet qualities. This might be explained with the longer material residence time in the barrel causing longer mixing times. This improves the binder distribution and allows the formation of more bridges between the powder particles. Figure 2 summarizes some of the found relationships between basic binder properties and the related granulation results. It was established with dicalcium phosphate and mannitol as models for insoluble and soluble formulations respectively. It is important to remember that the binder effectiveness cannot be attributed to only one single binder characteristic but to an interplay of different properties.
Figure 2: Concise overview of the critical binder attributes influencing the binder effectiveness for a poorly and highly soluble formulation (from Vandevivere L., et al. 2021).
Starch-based wet granulation binder
Starches and pregelatinized starches
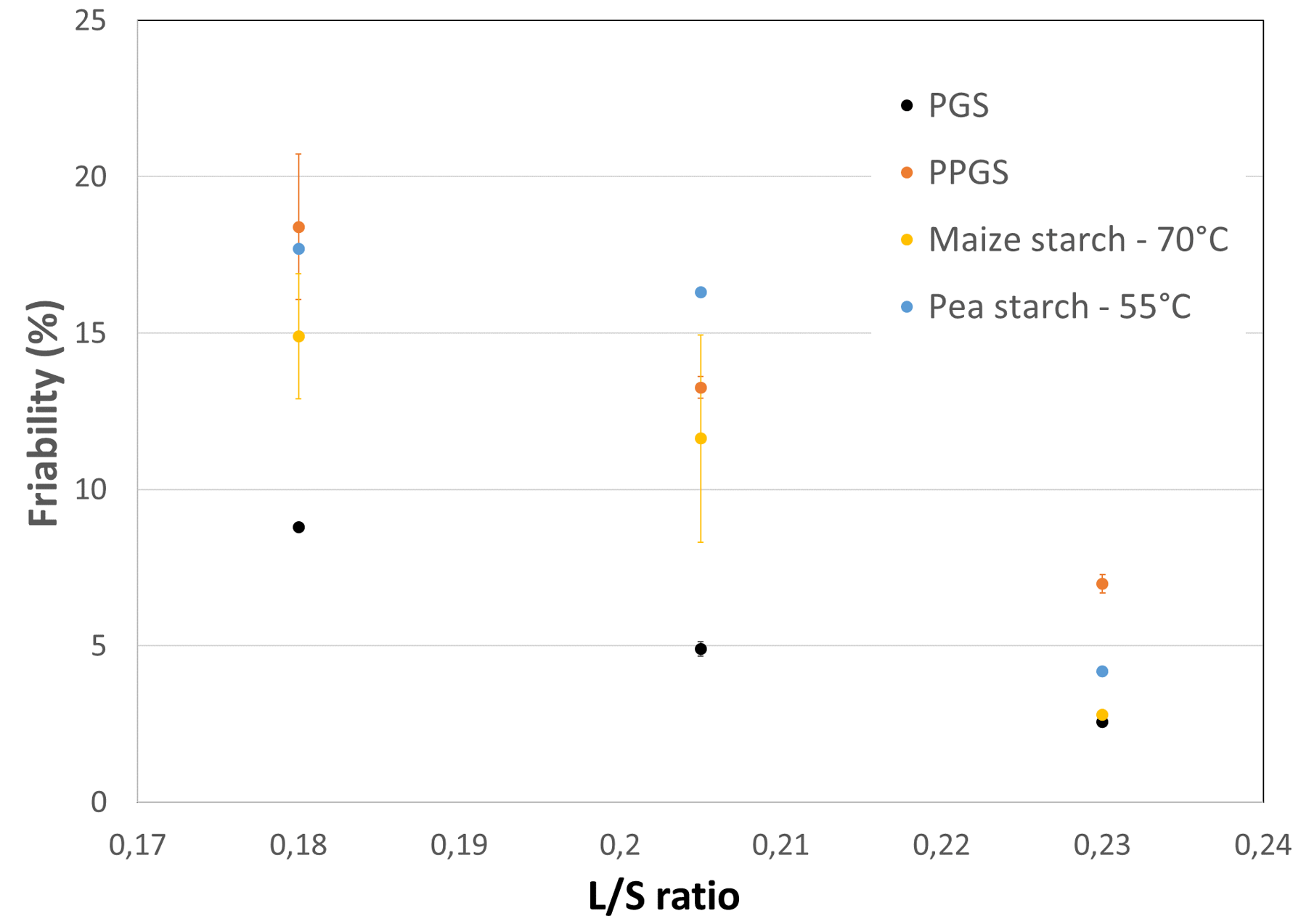
Granular starches are omnipresent excipients, serving either as wet granulation binder, disintegrant or glidant. The use of granular starch as WG binder requests a prior starch cooking step for its full activation. In continuous process, this cooking is only possible under very specific conditions. Of highest importance is to provide enough mechanical energy to assist the starch cooking, combined with higher process temperatures (depending on the gelatinization temperature of the starch). Systematic trials showed that starch gelatinization is only observed in insoluble systems and by using a screw with enhanced kneading zones. Globally, the obtained granule friability is rather low, caused by the high molecular weight of the starch polymer.
On the other side, the selection of appropriate granulation conditions permits to maintain granular starch in tablet formulations, e.g., as performing disintegrant, without risking any functionality loss due to a physical modification.
Figure 3: friability of dicalcium phosphate granulated with several starches at different L/S ratios. The recipe is 95% dicalcium phosphate and 5% starch (in dry blend). The temperature indicates the minimum temperature to obtain the binder activation of granular starches.
PGS = LYCATAB® PGS (fully pregelatinized starch); PPGS = LYCATAB® C (partially pregelatinized starch)
Pregelatinized starch acts always at lower L/S ratios (and room temperature) as binder, because there is no need for prior use of gelatinization. Partially pregelatinized starch can therefore be of interest in formulations, where this excipient type contributes both with its binding and disintegrant properties.
Maltodextrins and modified starches
Maltodextrins are obtained from partial hydrolysis of starch. They could be considered as fast activated binders with low viscosity and fit perfectly into hydrophilic and soluble systems. Compared with some modified starches, they might require higher L/S ratios for full activation, especially in poorly soluble systems.
The introduction of a chemical modification on the starch backbone can significantly change the wetting properties of the starch but also its plasticity in wetted state. Two starch derivatives with outstanding properties rank among the best-in-class WG binders.
Sodium starch octenylsuccinate starches (CLEARGUM® CO 01 and CO 03) own enhanced surfactant and wetting properties. Such binders could therefore be extremely easily activated, even at very low L/S ratios. Their use contributes to a significantly reduced water need and helps to shorten the drying times, hence increasing the productivity of the complete continuous production line. On the other hand, the process parameters should be very carefully selected to avoid an over granulation of the material. Such starches are also ideal for granulating hydrophobic materials, because they provide the lowest contact angles and reduce the energy barrier between liquid and solid, facilitating the spreading of the droplet over the powder.
Hydroxypropylated (HP) pea starch (LYCOAT® RS 720) has been identified as a universal WG binder, avoiding many industrial challenges. It is active over a very broad L/S ratio, contributing to the formation of granules with low friability, even varying the process conditions results in most cases into a monomodal particle size distribution. Wet granules, made with HP pea starch are more resistant against granule breakage induced by line and product transfer.
Summary
Implementation of continuous production processes is a new trend in the pharmaceutical industry. It enables a better control of the product quality, shorter production times and reduced production costs. However, such new processes present new constraints that require formulation optimization, more particularly by considering excipients with appropriate properties.
Starch and starch derivatives are valuable excipients for continuous wet granulation processes. Depending on their physical or chemical modification, they present varying hydration speed and surface energy as well as a 2-in-1 binder and disintegrant properties. They thus offer a wide range of functionalities fitting with APIs ranging from poorly to highly water soluble.
References
1. Dhenge, R.M.; Cartwright, J.J.; Hounslow, M.J.; Salman, A.D. Twin screw wet granulation: Effects of properties of granulation liquid. Powder Technology 2012, 229, 126–136
https://doi.org/10.1016/j.powtec.2012.06.019
2. Djuric, D.; Kleinebudde, P. Impact of screw elements on continuous granulation with a twin-screw extruder. Journal of Pharmaceutical Sciences 2008,97, 4934–4942
https://doi.org/10.1002/jps.21339
3. Menth, J.; Maus, M.; Wagner, K.G. Continuous twin screw granulation and fluid bed drying: a mechanistic scaling approach focusing optimal tablet properties, International Journal of Pharmaceutics 2020, 586, 119509
https://doi.org/10.1016/j.ijpharm.2020.119509
4. Pauli, V.; Kleinebudde, P.; Krumme, M. Predictive Model-Based Process Start-Up in Pharmaceutical Continuous Granulation and Drying, Pharmaceutics 2020, 12, 67
https://doi.org/10.3390/pharmaceutics12010067
5. Portier C, Vervaet C., Vanhoorne V. Continuous Twin Screw Granulation: A Review of Recent Progress and Opportunities in Formulation and Equipment Design. Pharmaceutics 2021, 13 (5), 668, DOI
https://doi.org/10.3390/pharmaceutics13050668
6. Köster, C.; Pohl, S.; Kleinebudde, P. Evaluation of Binders in Twin-Screw Wet Granulation, Pharmaceutics 2021, 13, 241.
https://doi.org/10.3390/pharmaceutics13020241
7. Kyttä, K.M.; Lakio, S.; Wikström, H.; Sulemanji, A.; Fransson, M.; Ketolainen, J.; Tajarobi, P. Comparison between twin-screw and high-shear granulation - The effect of filler and active pharmaceutical ingredient on the granule and tablet properties, Powder Technology 2020, 376, 187–198
https://doi.org/10.1016/j.powtec.2020.08.030
8. Roggo, Y.; Pauli, V.; Jelsch; M. Pellegatti, L.; Elbaz, F.; Ensslin, S.; Kleinebudde, P.; Krumme, M. Continuous manufacturing process monitoring of pharmaceutical solid dosage form: A case study. Journal of Pharmaceutical and Biomedical Analysis 2020, 179, 11297
https://doi.org/10.1016/j.jpba.2019.112971
9. Vandevivere, L.; Portier, C.; Vanhoorne, V.; Häusler, O.; Simon, D.; De Beer, T. Vervaet, C. Native starch as in situ binder for continuous twin screw wet granulation. International Journal of Pharmaceutics 2019, 571, 118760
https://doi.org/10.1016/j.ijpharm.2019.118760
10. Vandevivere, L.; Vangampelaere, M.; Portier, C.; de Backere, C.; Häusler, O.; De Beer, T. Vervaet, C.; Vanhoorne, V. Identifying Critical Binder Attributes to Facilitate Binder Selection for Efficient Formulation Development in a Continuous Twin Screw Wet Granulation Process, Pharmaceutics 2021, 13, 210.
https://doi.org/10.3390/pharmaceutics13020210
11. Vandevivere L.; Wijmeersch, E.; Häusler, O.; De Beer, T. Vervaet, C.; Vanhoorne, V. The effect of screw configuration and formulation variables on liquid requirements and granule quality in a continuous twin screw wet granulation process, Journal of Drug Delivery Science and Technology 2022, 68 103042
https://doi.org/10.1016/j.jddst.2021.103042
12. Vanhoorne, V.; Kumar, A. Advances in Twin-Screw Granulation, Pharmaceutics 2022, 14, 46.
https://doi.org/10.3390/pharmaceutics14010046
13. Van Snick, B.; Dhondt, J.; Pandelaere, K.; Bertels, J.; Mertens, R.; Klingeleers, D.; Di Pretoro, G.; Remon, J.-P.; Vervaet, C.; De Beer, T.; Vanhoorne, V. A multivariate raw material property database to facilitate drug product development and enable in-silico design of pharmaceutical dry powder processes, International Journal of Pharmaceutics 549 2018, 415–435
https://doi.org/10.1016/j.ijpharm.2018.08.014
14. Wahlich, J. Review: Continuous Manufacturing of Small Molecule Solid Oral Dosage Forms, Pharmaceutics 2021, 13, 1311.
https://doi.org/10.3390/pharmaceutics13081311
® Registered trademark(s) of Roquette Frères.
The information contained in this document is to the best of our knowledge true and accurate, but all instructions, recommendations or suggestions are made without any guarantee. Since the conditions of use are beyond our control, we disclaim any liability for loss and/or damage suffered from use of these data or suggestions. Furthermore, no liability is accepted if use of any product in accordance with these data or suggestions infringes any patent. No part of this document may be reproduced by any process without our prior written permission. For questions about a product’s compliance with additional countries’ standards not listed above, please contact your local Roquette representative.