LYCATAB® C - PREGELATINIZED MAIZE STARCH
LYCATAB®
®Registered Trademark(s) of Roquette Frères
- The excipient with the fastest disintegration, providing higher productivity
- Adapted to hard capsule filling
Partially pregelatinized starch
Applications
- 1. Solid Forms - Tablets
- Swallowable Tablets
- 2. Solid Forms - Capsules
- Hard Capsules Fill
- 5. Other Solid Forms
- Stick Packs and sachets
Functional properties
- Formulations Aids
- Disintegrants & Super Disintegrants
- Fillers and Binders for Direct Compression
- Fillers and Binders for Roller Compaction
Physical and chemical properties
- Formulation & Manufacturing
- Consistent drug dissolution, uniform weight
- Physical Characteristics
- Easy-flowing powder
- General Properties
- Label-friendly
- Multicompendial
- Stability & Compliance
- pH-independent disintegration
Documents
Product Specification Sheet
Name
Region
Size
Download
LYCATAB® C
Asia, JP
469,49 Ko
Region : Asia, JP
469,49 Ko
LYCATAB® C
475,42 Ko
Region :
475,42 Ko
Safety Data Sheet
Name
Region
Language
Size
Download
LYCATAB® C
Oceania, AU
EN
266,54 Ko
Region : Oceania, AU
EN
266,54 Ko
LYCATAB® C
Europe, BE
EN
280,99 Ko
Region : Europe, BE
EN
280,99 Ko
LYCATAB® C
Asia, CN
EN
273,77 Ko
Region : Asia, CN
EN
273,77 Ko
LYCATAB® C
Europe, CH
EN
281,37 Ko
Region : Europe, CH
EN
281,37 Ko
LYCATAB® C
Europe, DE
EN
285,88 Ko
Region : Europe, DE
EN
285,88 Ko
LYCATAB® C
Europe, FI
EN
281,02 Ko
Region : Europe, FI
EN
281,02 Ko
LYCATAB® C
Europe, FR
EN
281,21 Ko
Region : Europe, FR
EN
281,21 Ko
LYCATAB® C
Americas, Asia, Oceania, Africa
EN
432,60 Ko
Region : Americas, Asia, Oceania, Africa
EN
432,60 Ko
LYCATAB® C
Americas, Asia, Oceania, Africa
EN
265,20 Ko
Region : Americas, Asia, Oceania, Africa
EN
265,20 Ko
LYCATAB® C
Americas, Asia, Oceania, Africa
EN
386,26 Ko
Region : Americas, Asia, Oceania, Africa
EN
386,26 Ko
LYCATAB® C
Europe, AT
DE
314,81 Ko
Region : Europe, AT
DE
314,81 Ko
LYCATAB® C
Europe, BE
DE
314,62 Ko
Region : Europe, BE
DE
314,62 Ko
LYCATAB® C
Americas, BR
PT
337,58 Ko
Region : Americas, BR
PT
337,58 Ko
LYCATAB® C
Europe, BE
NL
306,11 Ko
Region : Europe, BE
NL
306,11 Ko
LYCATAB® C
Asia, CN
ZH
463,52 Ko
Region : Asia, CN
ZH
463,52 Ko
LYCATAB® C
Europe, CH
DE
314,84 Ko
Region : Europe, CH
DE
314,84 Ko
LYCATAB® C
Europe, CH
IT
308,95 Ko
Region : Europe, CH
IT
308,95 Ko
LYCATAB® C
Europe, CH
FR
323,15 Ko
Region : Europe, CH
FR
323,15 Ko
LYCATAB® C
Europe, BE
FR
320,72 Ko
Region : Europe, BE
FR
320,72 Ko
LYCATAB® C
Europe, DK
DA
308,23 Ko
Region : Europe, DK
DA
308,23 Ko
LYCATAB® C
Europe, FI
FI
316,40 Ko
Region : Europe, FI
FI
316,40 Ko
LYCATAB® C
Europe, CZ
CS
345,12 Ko
Region : Europe, CZ
CS
345,12 Ko
LYCATAB® C
Europe, DE
DE
318,34 Ko
Region : Europe, DE
DE
318,34 Ko
LYCATAB® C
Europe, ES
ES
315,94 Ko
Region : Europe, ES
ES
315,94 Ko
LYCATAB® C
Americas, Asia, Oceania, Africa
ES
293,77 Ko
Region : Americas, Asia, Oceania, Africa
ES
293,77 Ko
Get in touch with our Technical Experts
Please feel free to contact our technical experts for support during the development process.
Technical data
| Synonyms | Pregelatinized Starch |
|---|---|
| CAS number | 9005-25-8 |
| Physical form or apperance | White or almost white powder |
| Application | LYCATAB® C partially pregelatinized starch is a direct compression excipient with filler/binder and disintegrant properties. It is used both in nutraceutical and pharmaceutical oral dosage forms, mainly for swallowable tablets and hard capsules. |
| Source | Maize |
| Teste/Odor | Odorless powder |
| Morphology |
|
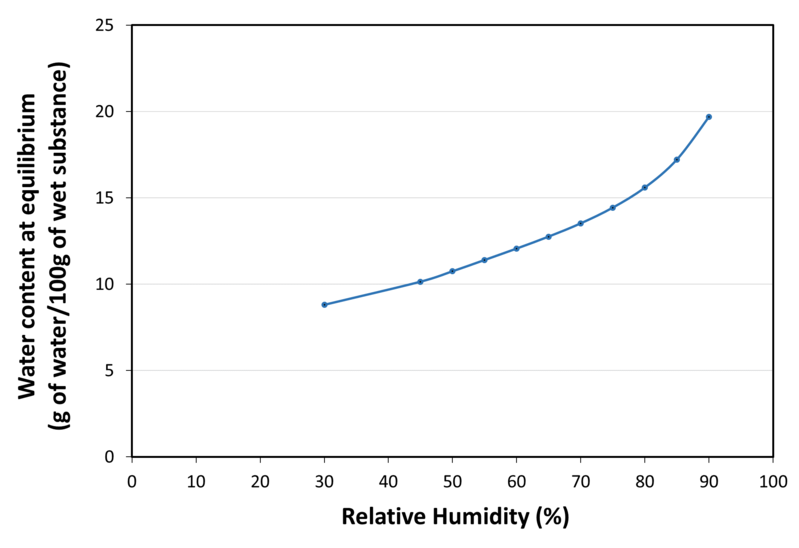
| Water sorption isotherm at 20°C |
|
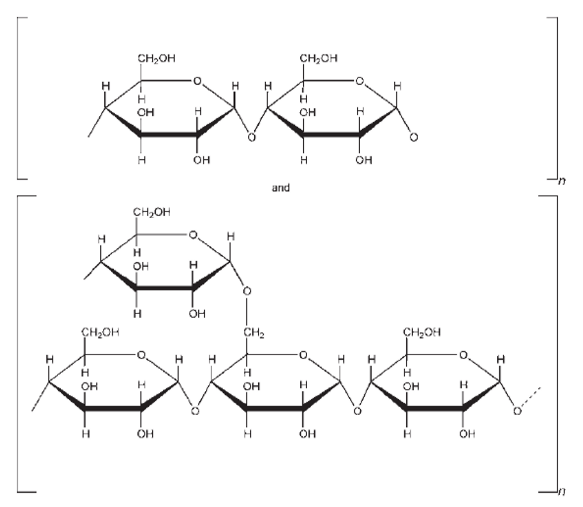
| Chemical Structure |
|
| Maximal Water content (LOD) | 14.00 |
| Solubility | Swells in cold water, practically insoluble in organic solvents |
| Average mean particle diameter | 90 |
| dv10 Particle size distribution | 35 |
| dv50 Particle size distribution | 90 |
| dv90 Particle size distribution | 166 |
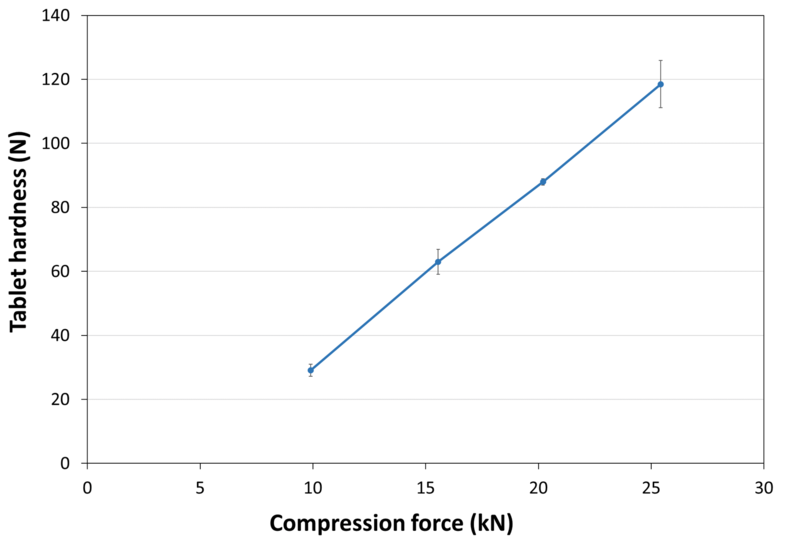
| Tablet Hardness |
|
| Experimental Conditions for Compression Behavior_Tablet Press | KORSCH XP 1 |
| Experimental Conditions for Compression Behavior_Production Speed | 20 tablets/min |
| Experimental Conditions for Compression Behavior_Tooling | Diameter 13 mm R13 concave |
| Experimental Conditions for Compression Behavior_Formuls | 100% LYCATAB® C |
| Experimental Conditions for Compression Behavior_Tablet Mass | 600 mg |
| Powder Flowability (according to Ph.Eur. 2.9.16, 10mm outflow opening) | 5 |
| Bulk Density (g/ml) | 0.63 |
| Tapped Density (g/ml) | 0.81 |
| True Density (g/ml) | 1.52 |
| Specific Surface Area (m²/g) | 0.30 |
| Angle of Repose (°) | 30 |