NEOSORB® P 200 SD - SORBITOL
NEOSORB®
®Registered Trademark(s) of Roquette Frères
- Designed for direct compression
- Optimized to absorb liquid
Particle size mean diameter - 200 µm approx
Applications
- 1. Solid Forms - Tablets
- Chewable Tablets
- Swallowable Tablets
- Effervescent Tablets
- 5. Other Solid Forms
- Losenges
Functional properties
- Formulations Aids
- Fillers and Binders for Direct Compression
- Sensory Enhancers
- Bulk sweeteners
- Sweetening agent
Physical and chemical properties
- General Properties
- Label-friendly
- Health & Nutritional Benefits
- Sugar-free
- Gluten-free
- Non-cariogenic
Documents
Product Specification Sheet
Name
Region
Size
Download
NEOSORB® P200 SD
Asia, CN
478,98 Ko
Region : Asia, CN
478,98 Ko
NEOSORB® P200 SD
487,01 Ko
Region :
487,01 Ko
Safety Data Sheet
Name
Region
Language
Size
Download
NEOSORB® P200 SD
Europe, BE
EN
299,12 Ko
Region : Europe, BE
EN
299,12 Ko
NEOSORB® P200 SD
Europe, DE
EN
302,12 Ko
Region : Europe, DE
EN
302,12 Ko
NEOSORB® P200 SD
Europe, CH
EN
299,56 Ko
Region : Europe, CH
EN
299,56 Ko
NEOSORB® P200 SD
Americas, Asia, Oceania, Africa
EN
406,98 Ko
Region : Americas, Asia, Oceania, Africa
EN
406,98 Ko
NEOSORB® P200 SD
Americas, Asia, Oceania, Africa
EN
454,40 Ko
Region : Americas, Asia, Oceania, Africa
EN
454,40 Ko
NEOSORB® P200 SD
Europe, FR
EN
299,50 Ko
Region : Europe, FR
EN
299,50 Ko
NEOSORB® P200 SD
Europe, ES
EN
299,16 Ko
Region : Europe, ES
EN
299,16 Ko
NEOSORB® P200 SD
Europe, IT
EN
301,58 Ko
Region : Europe, IT
EN
301,58 Ko
NEOSORB® P200 SD
Americas, Asia, Oceania, Africa
EN
279,46 Ko
Region : Americas, Asia, Oceania, Africa
EN
279,46 Ko
NEOSORB® P200 SD
Europe, NL
EN
296,20 Ko
Region : Europe, NL
EN
296,20 Ko
NEOSORB® P200 SD
Europe, BE
NL
330,56 Ko
Region : Europe, BE
NL
330,56 Ko
NEOSORB® P200 SD
Europe, BE
FR
346,95 Ko
Region : Europe, BE
FR
346,95 Ko
NEOSORB® P200 SD
Europe, BE
DE
338,06 Ko
Region : Europe, BE
DE
338,06 Ko
NEOSORB® P200 SD
Europe, CH
DE
338,43 Ko
Region : Europe, CH
DE
338,43 Ko
NEOSORB® P200 SD
Europe, CH
IT
334,36 Ko
Region : Europe, CH
IT
334,36 Ko
NEOSORB® P200 SD
Americas, BR
PT
358,71 Ko
Region : Americas, BR
PT
358,71 Ko
NEOSORB® P200 SD
Europe, CH
FR
347,61 Ko
Region : Europe, CH
FR
347,61 Ko
NEOSORB® P200 SD
Europe, CZ
CS
374,98 Ko
Region : Europe, CZ
CS
374,98 Ko
NEOSORB® P200 SD
Americas, Asia, Oceania, Africa
PT
321,80 Ko
Region : Americas, Asia, Oceania, Africa
PT
321,80 Ko
NEOSORB® P200 SD
Europe, ES
ES
341,47 Ko
Region : Europe, ES
ES
341,47 Ko
NEOSORB® P200 SD
Europe, FR
FR
347,15 Ko
Region : Europe, FR
FR
347,15 Ko
NEOSORB® P200 SD
Europe, DE
DE
343,19 Ko
Region : Europe, DE
DE
343,19 Ko
NEOSORB® P200 SD
Americas, Asia, Oceania, Africa
RU
358,98 Ko
Region : Americas, Asia, Oceania, Africa
RU
358,98 Ko
NEOSORB® P200 SD
Europe, IT
IT
336,32 Ko
Region : Europe, IT
IT
336,32 Ko
NEOSORB® P200 SD
Americas, Asia, Oceania, Africa
ES
316,96 Ko
Region : Americas, Asia, Oceania, Africa
ES
316,96 Ko
Get in touch with our Technical Experts
Please feel free to contact our technical experts for support during the development process.
Technical data
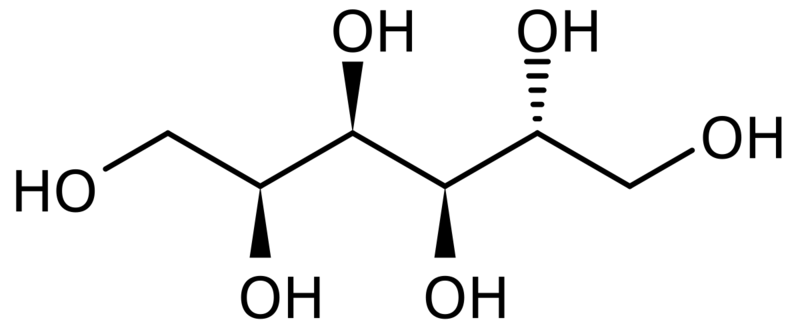
| Synonyms | D-Glucitol |
|---|---|
| CAS number | 50-70-4 |
| Physical form or apperance | White or almost white crystalline powder |
| Application | NEOSORB® P 200 SD sorbitol is a direct compression excipient. It is a non-cariogenic and non-acidogenic sugar-free sweetener that is suitable for chewable, suckable or effervescent tablets |
| Teste/Odor | Slightly sweet (sweetening power about 50-60% that of sucrose) with cooling effect due to high dissolution enthalpy in water (111 J/g) |
| Morphology |
|
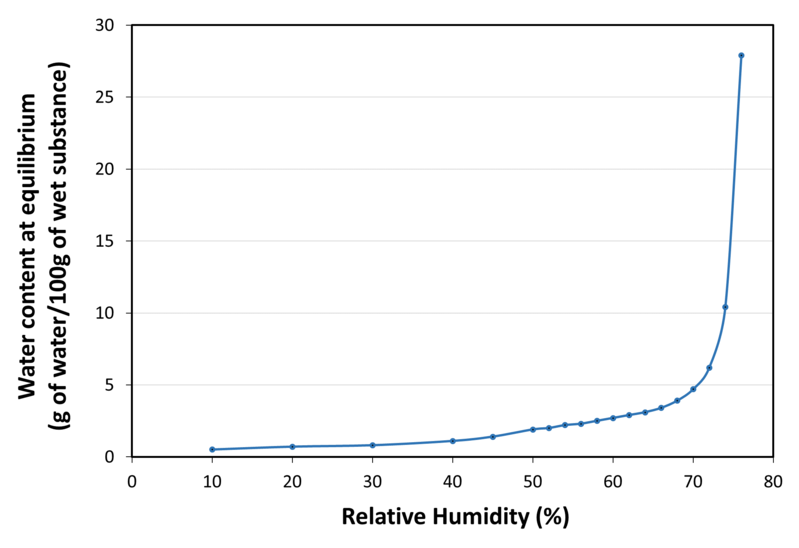
| Water sorption isotherm at 20°C |
|
| Chemical Structure |
|
| Average molecular weight | 182.2 g/mol |
| Nominal Water content (LOD) | 0.50 |
| Maximal Water content (LOD) | 1.50 |
| Solubility | Very soluble in water (235 g/100 ml of water at 25°C), practically insoluble in ethanol (96%) |
| Minimum melting temperature | 95 °C |
| Maximum melting temperature | 98 °C |
| Average mean particle diameter | 200 |
| dv10 Particle size distribution | 85 |
| dv50 Particle size distribution | 180 |
| dv90 Particle size distribution | 285 |
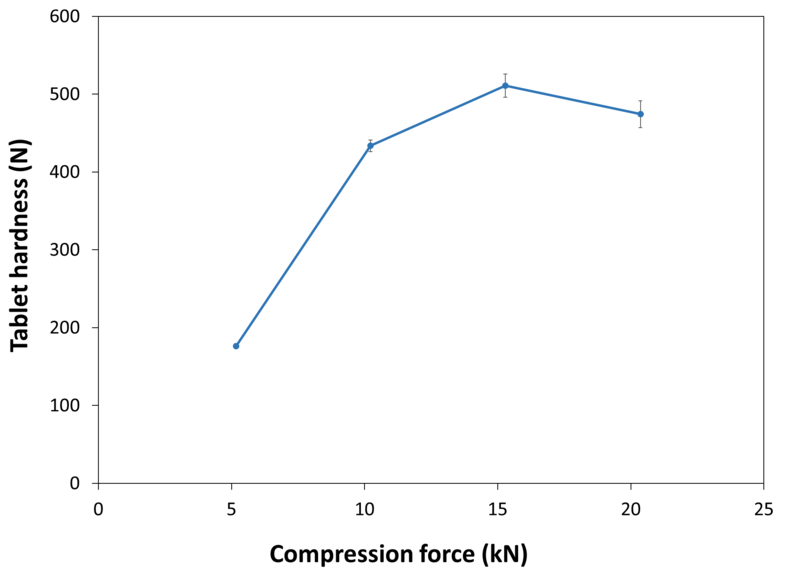
| Tablet Hardness |
|
| Experimental Conditions for Compression Behavior_Tablet Press | STYLCAM 200R |
| Experimental Conditions for Compression Behavior_Production Speed | 40 tablets/min (linear punch velocity: 152 mm/s; simulated rotary press speed: 250000 tablets/hour) |
| Experimental Conditions for Compression Behavior_Tooling | Diameter 10 mm R9 concave |
| Experimental Conditions for Compression Behavior_Formuls | 99.3% NEOSORB® P 200 SD / 0.7% magnesium stearate |
| Experimental Conditions for Compression Behavior_Tablet Mass | 400 mg |
| Powder Flowability (according to Ph.Eur. 2.9.16, 10mm outflow opening) | 7 |
| Bulk Density (g/ml) | 0.43 |
| Tapped Density (g/ml) | 0.51 |
| True Density (g/ml) | 1.51 |
| Specific Surface Area (m²/g) | 2.00 |
| Angle of Repose (°) | 32 |