Binder for continuous wet granulation: case of insoluble system
Published June 10, 2026
Authors
Olaf Haeusler
Pharm.D. - Global Technical Application Specialist, Roquette
Nicolas Descamps
Manager of the Functional Properties Analytical Laboratory, Roquette R&D
Introduction
A successful wet granulation procedure requests the use of appropriate granulation binder, usually cold-water soluble polymers. A careful binder selection is needed, especially for systems that are completely insoluble in water (such as dicalcium phosphate). In fact, such systems (excipients and/or APIs) cannot contribute to the granules formation with their own solubility (and hence binding capacity). In addition, a good wettability of the powders surface by the wetted binder is mandatory to guarantee the efficacy of the process.
In such particular insoluble systems, the obtained granules properties can directly be attributed to the used binder and its specific attributes. Studying them contributes both to a better knowledge of the binder itself, as well as a better understanding of the key characteristics to consider for an optimal designed formulation.
When using the binder as powder, the injection of water into the granulating device (always fed with the dry powder blend) triggers the granulation process. Water activates the binder by wetting to obtain its hydrated state, before it is spread on the solid particles. This activation process depends on numerous polymer properties, studied in this work. One important process parameter for monitoring the granulation efficiency is the needed liquid/solid ratio (L/S ratio). It indicates the water need to obtain the optimal binder expression. The L/S-ratio gives valuable details about the use characteristics of each binder.
This study makes the link between the binder properties and the quality of the obtained granules. The quality of the granules is here assessed by their friability after drying. By experience, a friability value of the granules lower than 30% indicates a sufficient granulation process (Keleb 2002, Portier 2020, Vanhoorne 2016).
Materials and Methods
Materials
Anhydrous dicalcium phosphate (DCP) (Calipharm® A, Innophos, Chicago Heights, USA) is granulated via dry binder addition, activated with water. The binders are:
Hydroxypropyl (HP) pea starch (LYCOAT® RS 720), Roquette Frères, France
Sodium starch octenyl succinate (CLEARGUM® CO 03), Roquette Frères, France
Polyvinylpyrrolidone (PVP) K30 (Kollidon® K30), BASF, Germany
Methods
Binders were used at a concentration of 5% and were introduced via a dry-mixing step before granulation.
All wet granulation binders were characterized with various physical methods. Powders were analyzed as follows: particle size distribution, wettability by water (using drop shape analyzer applying the sessile drop method on tablets made of pure binder), specific surface area and water binding capacity (g of water/g of powder). Aqueous binder solutions (8% in water) were evaluated on the following criteria: wettability of solid surface (using drop shape analyzer applying the sessile drop method on DCP tablets and Teflon and PTFE surfaces), surface tension and viscosity.
The granulation experiments were performed using the twin screw granulator of a continuous from-powder-to-tablet line (ConsiGmaTM-25 system, GEA Pharma Systems, Wommelgem, Belgium). This continuous granulator consists of two co-rotating screws with a length-to-diameter (L/D) ratio of 20/1. Screw speed and throughput were fixed at 300 rpm and 20 kg/h.
After tray drying, the granules’ friability was measured using a friabilator, at a speed of 25 rpm for 10 min, by subjecting 10 g of granules together with 200 glass beads (mean diameter 4 mm) to falling shocks. The percentage of created fines (under 250 µm) was used to calculate the friability.
All data were treated via a principal component analysis SIMCA® 16 software (Sartorius Stedium Biotech, Umeå, Sweden). A score scatter plot of principal component analysis (not shown) permits to cluster binders as a function of their effectiveness.
Results
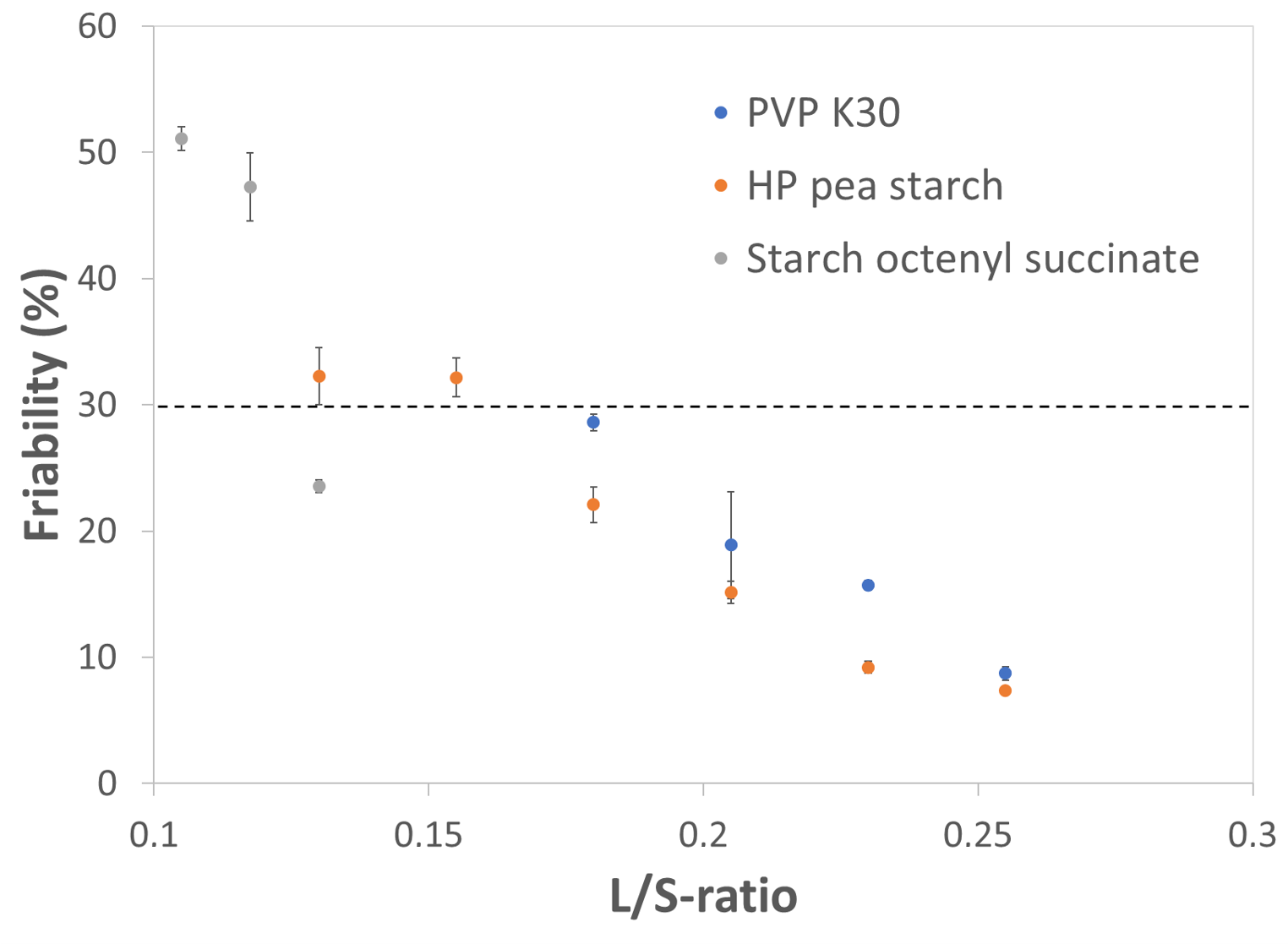
Significant differences in binder effectiveness are observed, considering the needed L/S-ratios to obtain an acceptable granule friability (≤30%). Figure 1 shows the binder efficiency (expressed as friability) as a function of the L/S ratio. Starch octenyl succinate always resulted at rather low L/S-ratios into acceptable low granule friability, compared with the other binders. It could therefore be considered as more effective. Starch octenyl succinate worked with typical L/S-ratios of (0.1175–0.1300) whereas the other binders were active at intermediate L/S-ratios (0.155–0.180). Some other binders such as maltodextrins (not displayed) are only active at rather high L/S ratios > 0.22.
Figure 1. Granule friability as a function of binder type and L/S-ratio. The dotted line represents a granule friability of 30%, set as maximum threshold to identify granules having sufficient strength.
It appears from the score scatter plot that the viscosity values of binder solutions are negatively correlated with the dissolution kinetics and the initial wetting of the powder.
Binder active at medium L/S-ratios (0.155–0.180) (PVP K 30 and HP starch) present intermediate properties – compared to all tested binders in the paper – considering the viscosity, initial wetting of the powder, surface tension, dissolution kinetics and wettability of hydrophobic surfaces. These binders are also characterized by having slower dissolution kinetics, which suggests swelling of the binders. In any case, HP starch LYCOAT® RS 720 results in good granulation over a very broad L/S-ratio range so that it could be considered as a versatile wet granulation binder, adapting to a multitude of application requests.
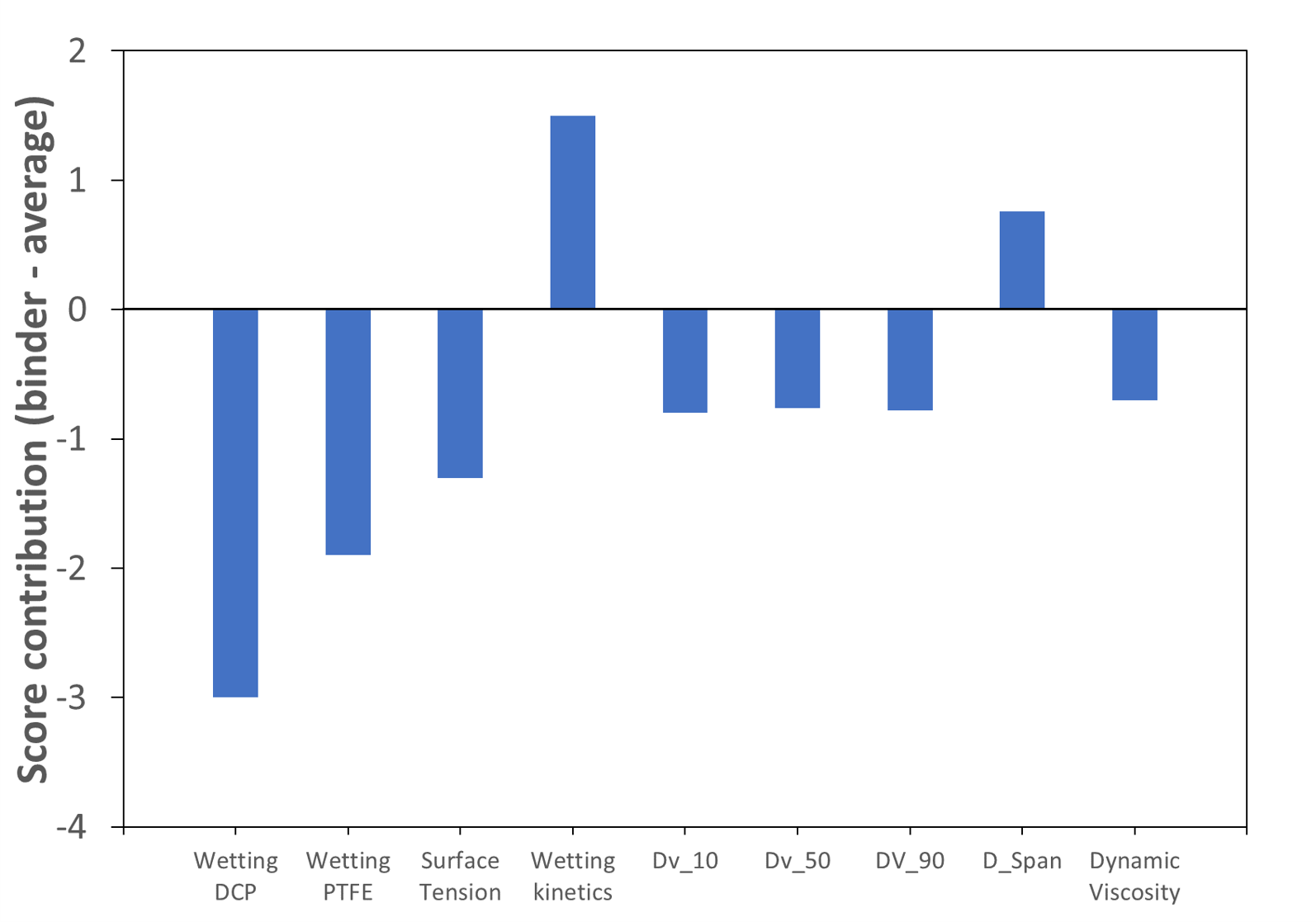
On the other hand, starch octenyl succinate shows fast auto-hydration combined with a good wetting capacity of different surfaces, low surface tension and small particle size. In addition, this binder operates at low L/S ratios. It becomes therefore rapidly activated and wets efficiently the DCP particles. Furthermore, the low surface tension of starch octenyl succinate solutions indicated a low energy barrier between liquid and air, facilitating the spreading of the droplet over the powder. Figure 2 shows the individual contribution of selected binder properties to its granulation efficacy. The high effectiveness at low L/S-ratios is linked to its fast wetting which leads to a rapid activation of the binder (positive correlation with the wetting kinetics, represented here by the spreading kinetics over 30s of a water droplet on a tablet made of pure binder). Afterwards, the activated binder could efficiently wet the DCP particles (wetting DCP and wetting PTFE attributes represent here the contact angles between the binder solution and DCP or PTFE surfaces). Compared to the other binders, starch octenyl succinate resulted in the lowest contact angle measured on DCP tablets with 32.8°.
Figure 2. Score contribution plot of starch octenyl succinate CLEARGUM® CO 03. Wetting DCP and PTFE represent here the contact angles between the binder solution and DCP or PTFE surfaces. Wetting kinetics represents here the spreading kinetics over 30s of a water droplet on a tablet made of pure binder.
Conclusion
The dry addition of wet granulation binder is possible, also for an insoluble powder system.
Studying the L/S –ratios in an insoluble model is an interesting tool to evaluate binder properties. It clearly shows that higher binder effectiveness is correlated with good wetting properties of the powder bed. The performance of wettability measurements in an early formulation development stage is therefore considered highly important.
In the present study, CLEARGUM® CO 03 demonstrates very good ability for being used as binder in a continuous granulation process. Its quick hydration capacity associated with a good wettability of hydrophobic surfaces make it a particularly suitable binder for such a process.
For more details, see the following article:
L. Vandevivere; P. Denduyver; C. Portier; O. Häusler; T. De Beer; C. Vervaet; V. Vanhoorne
Influence of binder attributes on binder effectiveness in a continuous twin screw wet granulation process via wet and dry binder addition
International Journal of Pharmaceutics, Volume 585, 30 July 2020, 119466
https://doi.org/10.1016/j.ijpharm.2020.119466
Further reading:
Keleb, E.I.; Vermeire, A.; Vervaet, C.; Remon, J.P. Continuous twin screw extrusion for the wet
granulation of lactose. Int. J. Pharm. 2002, 239, 69–80,
https://doi.org/10.1016/S0378-5173(02)00052-2.
Portier, C.; Pandelaere, K.; Delaet, U.; Vigh, T.; Di Pretoro, G.; De Beer, T.; Vervaet, C.;
Vanhoorne, V. Continuous twin screw granulation: A complex interplay between formulation
properties, process settings and screw design. Int. J. Pharm. 2020, 576, 119004,
https://doi.org/10.1016/j.ijpharm.2019.119004.
Vanhoorne, V.; Janssens, L.; Vercruysse, J.; De Beer, T.; Remon, J.P.; Vervaet, C. Continuous twin
screw granulation of controlled release formulations with various HPMC grades. Int. J. Pharm.
2016, 511, 1048–1057,
https://doi.org/10.1016/j.ijpharm.2016.08.020.
Willecke, N.; Szepes, A.; Wunderlich, M.; Remon, J.P.; Vervaet, C.; De Beer, T.
Identifying overarching excipient properties towards an in-depth understanding of process and product performance for continuous twin-screw wet granulation.
Int. J. Pharm. 522 (2017), 234–247.
https://doi.org/10.1016/j.ijpharm.2017.02.028.
El Hagrasy, A.S.; Hennenkamp, J.R.; Burke, M.D.; Cartwright, J.J.; Litster, J.D.
Twin screw wet granulation: Influence of formulation parameters on granule properties and growth behavior.
Powder Technol. 238, (2013) 108–115.
https://doi.org/10.1016/j.powtec.2012.04.035
Dhenge, R.M.; Cartwright, J.J.; Hounslow, M.J.; Salman, A.D.; 2012 Twin screw wet
granulation: Effects of properties of granulation liquid.
Powder Technol. 229, 126–136.
https://doi.org/10.1016/j.powtec.2012.06.019.