Comparison of Different Sorbitol Sorbitan Solutions as Gelatin Plasticizer
Published May 28, 2026
Authors
Olaf Haeusler
Pharm.D. - Global Technical Application Specialist, Roquette
Gregory Le Bihan
Global Pharma Application Support, Roquette
Laurent Dambrine
Chemistry Research Technician, Roquette
Sandra Bigot
Chemical Engineer, Roquette
Introduction
Soft capsules are a popular dosage form for dispensing active pharmaceutical or nutraceutical materials. The shell material is frequently composed of gelatin, softened with considerable amount of plasticizer. They are especially adapted for liquids and for the formulation of hardly soluble drug substances.
In many cases, glycerin serves as main or as sole gelatin plasticizer. However, whenever the liquid fill material could absorb water, glycerin is not the premium formulation choice. In fact, some fills such as drug solutions in PEG or self-emulsifying systems do absorb the plasticizer and change therefore the properties of the capsule shell during storage.
An alternative plasticizer with higher molecular weight could limit its migration inside the capsule. Typical formulation strategies to encapsulate sensitive fill materials relying therefore on sorbitol plus glycerin blends or on the use of sorbitol-sorbitan solutions. Such excipients have lower mobility from the capsule shell, but they do not allow to reach the same plasticization efficacy than that of glycerin. It is known from prior studies that the relative composition of plasticizer modulates its interaction with gelatin.1, 2
Scientific assessments suggest that 1,4-sorbitan has a direct interaction with gelatin, resulting in a strong plasticizing effect. Sorbitol, on the other hand, is considered an indirect plasticizer, and other components of commercial sorbitol – sorbitan solutions might not have any plasticizing effect.
OBJECTIVES
A new experimental sorbitol-sorbitan solution (POLYSORB® 2.0, Roquette Frères, France), with an optimized composition, was tested for its capacity to plasticize gelatin. The commercial plasticizer POLYSORB® 85/70/00 (sorbitol-sorbitan solution, Roquette Frères, France) and glycerin (Arcane, France) were used as benchmarks.
Main attention was paid on important application parameters such as the flexibility of the capsules and its drying speed. Possible improvements of the efficiency of sorbitol-sorbitan solutions were monitored.
Table 1 summarizes the relative composition of POLYSORB® 85/70/00 and POLYSORB 2.0.
Table 1. Typical relative mass compositions of POLYSORB® 85/70/00 and POLYSORB 2.0.
| POLYSORB® 85/70/00 | POLYSORB® 2.0 | |
| Water content | 15% | 27.5% |
| Sorbitol content (in dry substance) | 40% | 30% |
| 1,4-sorbitan (in dry substance) | 26% | 55% |
| Other sorbitans (in dry substance) | 12% | 14% |
| Other polyols, eg., maltitol and mannitol (in dry substance) | 22% | 1% max. |
The significantly increased content in sorbitans should contribute to a better plasticizing capacity.
Materials and Methods
Materials
Gelatin 160 Bloom, bovine from Gelita GmbH (Eberbach, Germany)
Plasticizer:
- POLYSORB® 85/70/00 partially dehydrated sorbitol liquid (Roquette Frères, Lestrem, France)
- POLYSORB® 2.0, experimental product (Roquette Frères, Lestrem, France)
- Glycerin (vegetal) (Arcane, Aubagne, France)
As liquid fill material served PEG 400 (Merck, Darmstadt Germany)
Methods
Capsule production
Gelatin, plasticizer and water were heated at a temperature of 60°C to obtain a homogenous preparation before its transfer to a Changsung SRD line. The encapsulation was done using # oval 12 die rolls reaching a fill of 550 mg PEG 400. Capsules were dried in a 25% relative humidity / 20°C environment, first for 2h in a tumble dyer and later on open trays placed in a climatic chamber.
Capsule recipe
Table 2. Global recipe of soft capsules
| Material | Concentration (%) |
| Gelatin 160 Bloom | 39 % |
Plasticizer (exclusively one)
| 21 % |
| Water | 40 % |
Characterization of soft capsules
Capsule drying speed
Capsules were stored/dried at 20°C/25% relative humidity. Their weight changes were recorded daily. The starting point for the weight measurement was just after the tumbler drying (for 2h).
Capsule hardness
The hardness of capsules during drying was measured with an Instron texturometer. The needed force to press a 5 mm flat cylinder for a depth 2 mm was measured.
Capsule disintegration time
The capsule disintegration times were measured with a Sotax DT50 (Sotax AG Aesch, Switzerland).
Results
All tested plasticizers permitted the easy production of clear soft capsules without defects or leakage (see figure 1). They are stable for at least one year, when stored under normal room conditions.
Figure 1. Soft capsules plasticized with A) glycerin and B) POLYSORB® 2.0.
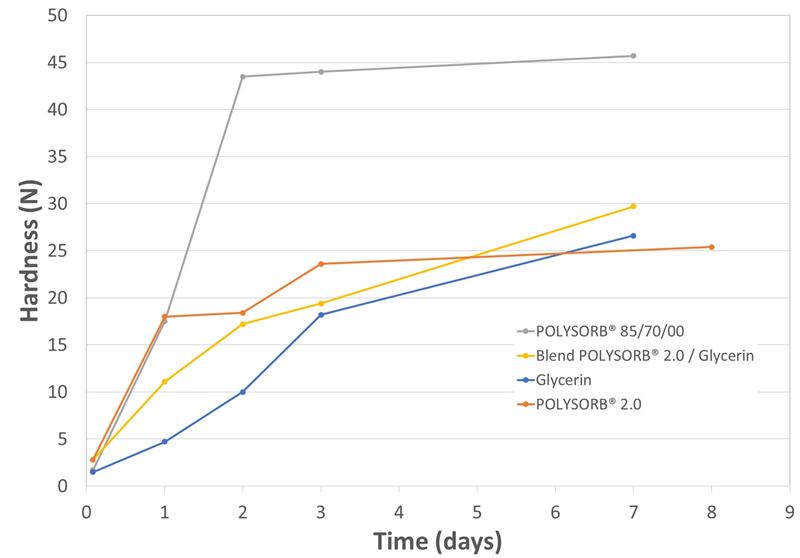
Figure 2. Evolution of the capsules' hardness during the drying process.
The evolution of the capsules’ hardness shows big differences between plasticizers (see figure 2).
The commercial POLYSORB® 85/70/00 resulted in rather rigid and hard capsules. Surprisingly, the tested new composition of sorbitol-sorbitan solution (POLYSORB® 2.0) has an improved capacity to soften gelatin. Its softening capacity is comparable with that of pure glycerin. In addition, the hardness of capsules plasticized with POLYSORB® 2.0 reaches a plateau only after three days of drying. This means that further desiccation will not contribute to increase capsules’ mechanical resistance.
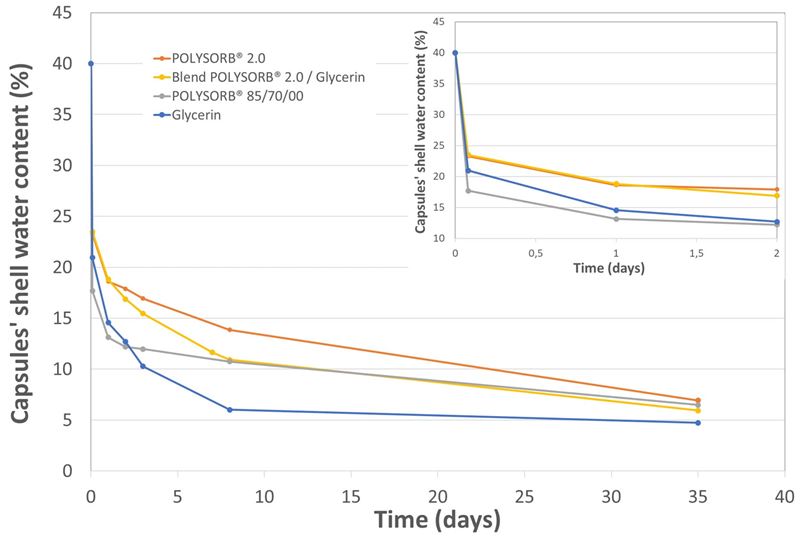
Figure 3 shows the evolution of the capsules’ shell water content upon drying. The first two hours of drying take place in the tumble dryer. The tray drying completes this initial drying step. Even though final water contents after 35 days are similar for all plasticizers, it appears that POLYSORB® 2.0 presents a slower drying kinetics.
However, contrary to glycerin, the combination of the results presented in figures 2 and 3 demonstrates that an extensive drying is not necessary for POLYSORB® 2.0 formula to reach mechanical resistance optimum value.
Figure 3. Water content of soft capsules’ shell containing different plasticizers.
Time = 0 : initial water content ; Time = 2 h : after drying in the tumble dryer ; Time > 2h : drying in climatic chamber at 20°C / 25 % relative humidity. The inset shows details of the first two days of drying.
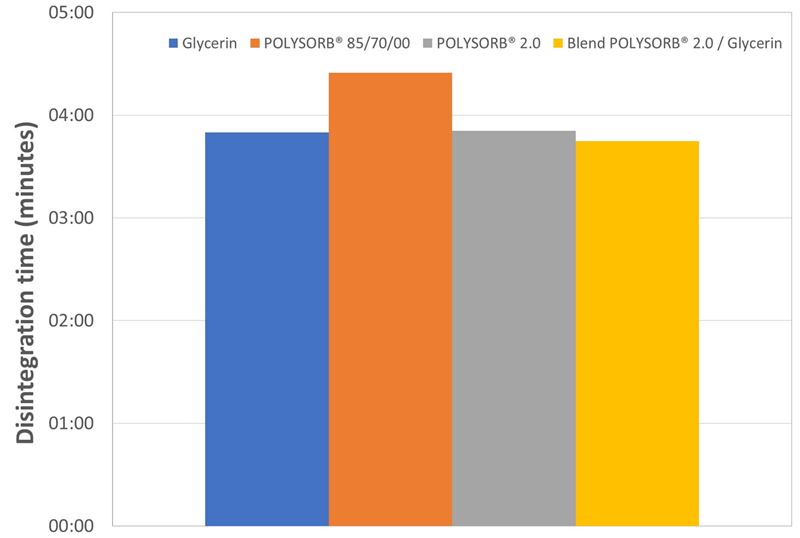
The disintegration times of all capsules, independent of the used plasticizer, are short. They are between 3 and 5 minutes (see figure 4).
Figure 4. Disintegration time of soft capsules plasticized with different plasticizers.
Conclusion
The nature of the plasticizer system has an important influence on capsule production, its properties and the capsules’ evolution during storage.
A sorbitol-sorbitan solution with a selected composition used as plasticizer results into more flexible gelatin capsules. It is an efficient substitute to glycerin for hydrophilic fills.
REFERENCES
- Reich G, Effect of sorbitol specification on structure and properties of soft gelatin capsules, Pharm. Ind. 58 (1996) 941–946
- Parissaux X, Häusler O, Eye on excipients; Tablets and capsules, April 2019
® Registered trademark(s) of Roquette Frères. The information contained in this document is to the best of our knowledge true and accurate, but all instructions, recommendations or suggestions are made without any guarantee. Since the conditions of use are beyond our control, we disclaim any liability for loss and/or damage suffered from use of these data or suggestions. Furthermore, no liability is accepted if use of any product in accordance with these data or suggestions infringes any patent. No part of this document may be reproduced by any process without our prior written permission. For questions about a product’s compliance with additional countries’ standards not listed above, please contact your local Roquette representative.